An Introduction to the Hexagonal Crystal System
- Jun 28, 2017
- 6 min read
I often compare the process of red beryl formation to the crystallization of rock candy from a super-saturated sugary solution -- except instead of water, Red Emeralds are born in burning, hot magma; a gift from lava.
Everything in nature attempts to reach a state of equilibrium, even from an energy perspective. A hot pan radiates heat to distribute temperature evenly throughout a room. An underground lava dome is like a continent-sized frying pan trying to shed energy into the environment as quickly and efficiently as possible. Crystal formation helps draw down this abundant thermal energy.
Volcanic gases are so hot and dense, they act like vaporized solids. This super-steam creates a mineral snowfall, with gases depositing precipitates continuously as they cool along their route of travel. To qualify as gemstones, the minerals deposited must first grow to be crystalline. To form a crystal, atoms repeatedly group into identical molecules which are then placed in a highly-ordered structure. The efficiency of this order directly impacts crystal quality. The organization of these molecules occurs on an imaginary grid called a Lattice.

Red lines through the repeating pattern of these tiles reveals a lattice.
Six oxygen atoms in Beryl form a hexagonal molecule, shaping the crystal.
The geometric figure of each molecule physically limits the number of ways it can be arranged, resulting in a finite number of angles at which crystal faces may form. Consequentially, crystals of the same mineral variety regularly produce structures in similar patterns called Crystal Habits. All crystals can be defined by the shape of their typical habit and taxonomically ordered in a Crystal System.
To determine which system a crystal belongs to, a detective must first locate the crystal's primary Axis. The two mathematical axes are the X-Axis and Y-Axis, but in a three dimensional sketch, we need a third Axis, the Z-Axis. In crystallography, these are relabeled as the A-Axis (X-Axis), the B-Axis (Y-Axis) and the C-Axis (Z-Axis).

Transformation from a two-dimensional graph into a three-dimensional (3D) model illustrating the orientation of red beryl.
Locating the C-Axis is important so gemologists, students and fans may accurately describe a specimen. For Emerald lapidaries, orienting a gemstone’s table perpendicular to the C-Axis often produces the best color and brilliance.
Since the C-Axis points "up" from the viewer's perspective, looking down the C-Axis is the same as looking down onto the "top" of a crystal. Take the geometric shape observed when viewing a crystal from this angle and rotate the specimen 360 degrees. How many times in that full rotation does the shape look identical to its appearance at the start? Answering this question defines a crystal's Rotational Symmetry.

In a full rotation, the hexagon appears similar six times
for a six-fold rotational symmetry.
When looking down onto the top of a standard red beryl crystal, a six-sided hexagon is observed. A full spin reveals the same shape six times, which means red beryl has six-fold rotational symmetry. This establishes that red beryl belongs to the Hexagonal System; no other crystal system has greater symmetry, and some will always consider the most to be the best. We will encounter many of the other six systems in later blog posts.

If the hexagon is stretched into a tower, the geometric shape is called a Trapezohedron. This crystal habit is called a "Prism" by mineral collectors.
When looking down onto a standard red beryl crystal, if a rectangle is observed, the detective made an error and is seeing one of the six crystal faces. Each face is typically offset 60 degrees from the faces on either adjoining side. The two crystal faces on opposite sides of a beryl crystal are a mirrored pair, because they are always oriented parallel to one other. The six faces of a beryl crystal create three sets of mirrored pairs.

Trapiches orient inclusions along the rotational axes in an attempt to maintain the highly-ordered molecular structures of their crystal form.
Looking down the C-Axis, imagine three lines which connect at the center-points of the mirrored pairs. These lines are physically present in a Trapiche Emerald, which arranges inclusions along these planes. The six-fold rotational symmetry of red beryl means there are six Axes on which a crystal may Rotate and remain symmetrical. The lines connecting the mirrored pairs represent the six Rotational Axes, as shown below.
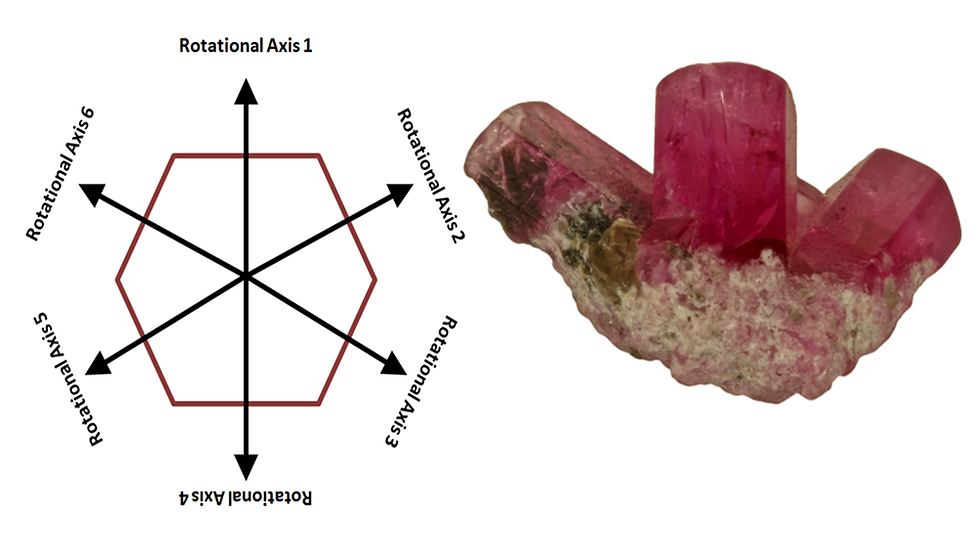
Precise 60 degree angles along rotational axes are maintained for the direction of growth in prism sprays of both green and red beryl specimens.
A disturbance during formation may cause a new crystal to form in the direction of a Rotational Axis. If a larger crystal shows preferential growth in one of these directions, the formation has "rotated" towards that axis. With a six-fold rotational axis, the Hexagonal System has the most rotation possibilities (options during formation) of all crystal systems.
Each Crystal System is broken down into Crystal Classes. The Hexagonal System has seven Crystal Classes. The names for these classes refer to the number of crystal faces each class can produce in its ultimate form. The number of possible crystal faces affects the potential complexities and variations of specimen forms in a mineral variety.
CRYSTAL CLASS # FACES
Hexagonal Pyramidal 6
Trigonal Dipyramidal 6
Hexagonal Dipyramidal 12
Hexagonal Trapezohedral 12
Dihexagonal Pyramidal 12
Ditrigonal Dipyramidal 12
Dihexagonal Dipyramidal 24
The mineral beryl forms in the Dihexagonal Dipyramidal class of the Hexagonal System, which is capable of producing the most natural crystal faces of any Class in any System. Some collectors will always consider the most to be the best, and favor this class.

This facet-grade Red Emerald specimen is a wonderful example
for the propagation of multiple crystals in a cluster.
The entire beryl family is filled with celebrities of the gemstone world, but the particular nature of Emerald formation causes their specimens to display a unique crystallography. Stressors in the environment often necessitate propagation of new crystals along rotational axes, encouraging cluster specimens which exhibit some of the most pronounced formations in the mineral world.
A beryl molecule is a cyclosilicate, with six oxygen atoms locked in hexagonal ring. As molecules of like kind begin to draw together, a larger mineral hexagon begins to appear. This hexagon is called the termination end, because this is where the specimen stops. A hexagon which grows into a short and tabular habit is referred to as a Wafer specimen.

Left: Digital Sketch of a Tabular Hexagon Wafer.
Technically, thousands of tiny independent beryl crystals are growing in unison, observed as hexagonal stacking. The edges and boundaries of crystals are flat and expand slowly over time, adding to their exterior like rings on a tree. Traces of this growth in parallel can be seen as angular zoning. Each of these are topics of future blog posts.
When a beryl grows under conditions favorable to long-term crystallization, hexagonal wafers continue to stack one upon another. If a specimen stacks high enough, its profile begins to appear like a tall mineral tower. A crystal in this habit is referred to as a Prismatic specimen.
Most prismatic specimens are produced in the Wah-Wah range. After the first 25 years of mining, the largest crystal discovered weighed only 30 carat and measured 22 millimeters in height by 12 millimeters in width and depth.

Two views of this 44.7 carat freestanding red beryl prism
show the meta-consistent, 60 degree angle of new crystal propagation.
Short crystals cannot cut gems deep enough for proper light return, and a beryl elongated to a point where it becomes needle-like is too long to be efficient. Some prism dimensions are better suited to faceting than others. The ideal Length-to-Width ratio for Emerald cut stones is 1.5:1 meaning length should be one and a half times longer than width. Prism proportions considered desirable depend on the crystal size required to produce an ideal faceted stone.

295 carat Pezzottaite from Bonham's and Benitoite Mirror Twin from the Whimsy Mine. "Stars" are impossibly rare, much less flawless examples.
When two crystals grow in the same direction but their geometric figures are offset, they are collectively referred to as a Mirror Twin, because they are reflections on a Mirror Plane within the Lattice. The typically triangular Benitoite produces Mirror Twin specimens with six-pointed terminations; these are hunted by collectors who describe the habit as a "Star of David". Mirror Twins result in twice as many faces -- the mirror twin of beryl is a Dihexagon with 12 crystal faces instead of six.
This is one step closer to the Dihexagonal Dipyramidal ultimate form!
In the ultimate form of beryl, the termination end is not flat, but instead has a pyramid termination. A Pyramid on the hexagonal end of beryl produces six additional crystal faces.

If a pyramid appeared on both the top and bottom terminations of a Dihexagon, the specimen would have Dipyramids, making the Dihexagonal Dipyramidal Ultimate Form.

Beryl are known to form phenomenally large prisms, so if nothing inhibits growth, an increase in crystal size usually appears, not a pyramid termination. Pyramids do not occur in full, but evidence of their presence seen in a modification of crystal habit is an outstanding prize in any collection.

Exceptionally coveted mirror plane and pyramidal modifications appear in Emerald as beveled (dome) and blunted point (pinacoid) alterations where crystal faces meet the termination end(s) and/or each other.
Although previously undocumented, these appear in the crystallography of Red Emerald as well as the Green! These structural similarities and much, MUCH MORE will be revealed in the weeks and months to come!







Comments